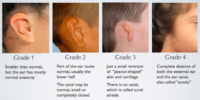
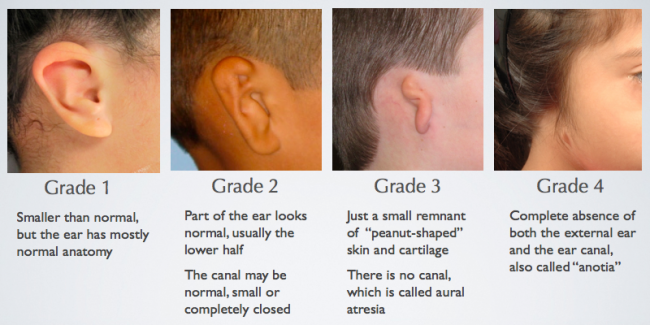
February 1st, 2023 — Pax’s Ear Fund is a hearing health reimbursement program offered through Ear Community that allows audiologists to be reimbursed for fitting/programming services when patients are denied coverage for this service by their insurance provider leaving them unable to pay the fee or high deductible at the time of the fitting/programming. This reimbursement program only covers patients born with microtia and/or aural atresia who wear Bone Anchored Hearing Devices/Bone Conduction Hearing Implant Systems. As a nonprofit organization that serves the microtia and aural atresia community, we ask that patients must have microtia and/or aural atresia to qualify as we have limited funds to work with for our community.
Pax’s Ear Fund is made possible by a sweet little boy named Pax who was born with microtia and atresia. Pax and his family know how important it is to help provide the gift of hearing to those in need. Ear Community is grateful for Pax’s Ear Fund! Pax’s fund gives Ear Community the opportunity to help give back to the Deaf and Hard of Hearing Community when helping members of the microtia and atresia community thrive.
It is Ear Community’s goal to help as many children and adults to hear their best as much as possible. Please check out the link below for program details and the reimbursement form: https://earcommunity.org/ear-community-launches-new-fitting-programming-reimbursement-program-for-audiologists/669596/
Thank you to AudiologyOnline Continued for sharing this program with audiologists everywhere!
Ear Community
For AO link, click here.